Bromine Ionic Or Covalent . Both types result in the stable electronic states associated with the noble gases. The first question we ask is if the compound is ionic or covalent? Covalent radius half of the distance between two atoms within a single covalent bond. Ionic bonds differ from covalent bonds. Compounds that do not contain ions, but instead consist of atoms bonded tightly. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. However, in covalent bonds, the electrons are shared between. Values are given for typical oxidation number and. Ionic compounds generally form from metals and nonmetals. That is, does it have ionic bonds, or covalent bonds?.
from www.youtube.com
The first question we ask is if the compound is ionic or covalent? Ionic bonds differ from covalent bonds. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. Ionic compounds generally form from metals and nonmetals. Both types result in the stable electronic states associated with the noble gases. That is, does it have ionic bonds, or covalent bonds?. However, in covalent bonds, the electrons are shared between. Covalent radius half of the distance between two atoms within a single covalent bond. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located. Compounds that do not contain ions, but instead consist of atoms bonded tightly.
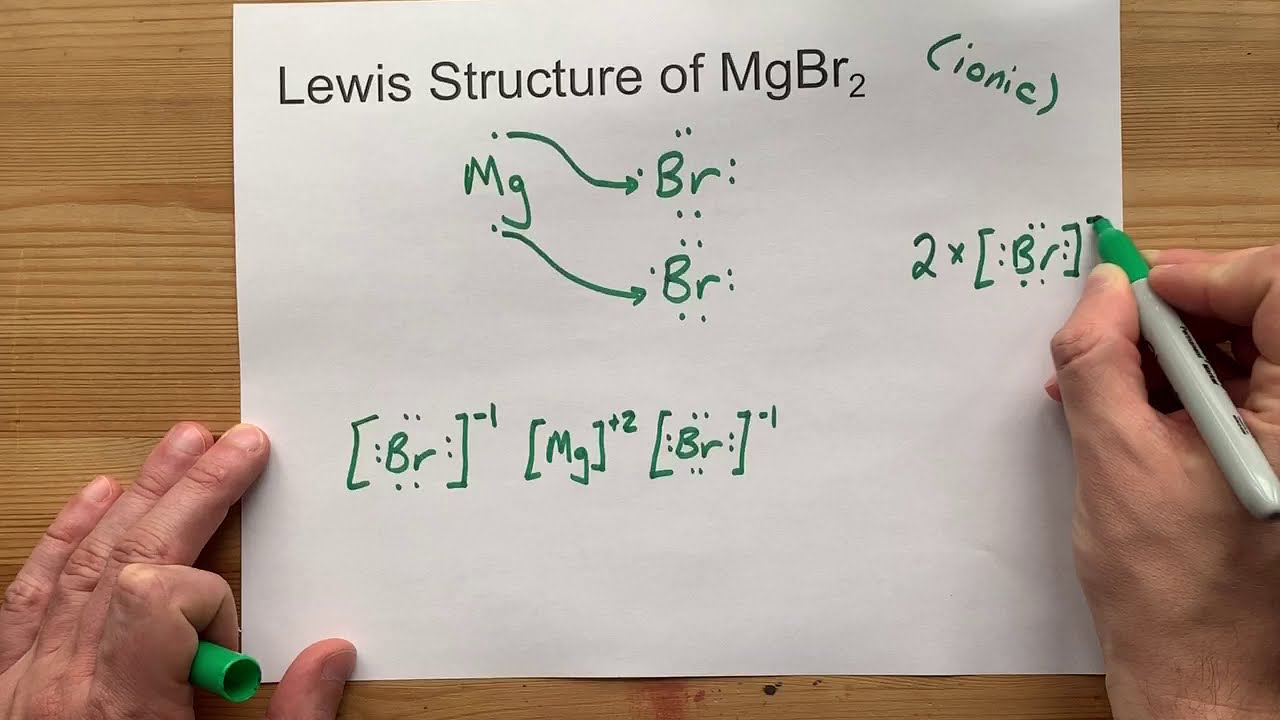
Draw the Lewis Structure of MgBr2 (magnesium bromide) YouTube
Bromine Ionic Or Covalent Compounds that do not contain ions, but instead consist of atoms bonded tightly. Both types result in the stable electronic states associated with the noble gases. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located. Values are given for typical oxidation number and. Ionic bonds differ from covalent bonds. Compounds that do not contain ions, but instead consist of atoms bonded tightly. However, in covalent bonds, the electrons are shared between. The first question we ask is if the compound is ionic or covalent? That is, does it have ionic bonds, or covalent bonds?. Ionic compounds generally form from metals and nonmetals. Covalent radius half of the distance between two atoms within a single covalent bond. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the.
From www.youtube.com
Lewis Structure of CsBr, caesium bromide YouTube Bromine Ionic Or Covalent Ionic compounds generally form from metals and nonmetals. Both types result in the stable electronic states associated with the noble gases. Values are given for typical oxidation number and. Covalent radius half of the distance between two atoms within a single covalent bond. However, in covalent bonds, the electrons are shared between. Compounds that do not contain ions, but instead. Bromine Ionic Or Covalent.
From www.thoughtco.com
Examples of Ionic Bonds and Ionic Compounds Bromine Ionic Or Covalent The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. Covalent radius half of the distance between two atoms within a single covalent bond. That is, does it have ionic bonds, or covalent bonds?. Ionic compounds generally form from metals and nonmetals. Values are. Bromine Ionic Or Covalent.
From chemnotcheem.com
Dot and cross diagrams of simple molecules Exam Questions Bromine Ionic Or Covalent Both types result in the stable electronic states associated with the noble gases. Values are given for typical oxidation number and. Covalent radius half of the distance between two atoms within a single covalent bond. The first question we ask is if the compound is ionic or covalent? However, in covalent bonds, the electrons are shared between. Ionic compounds generally. Bromine Ionic Or Covalent.
From www.teachoo.com
[Class 10] Differentiate between Ionic bond & Covalent bond Teachoo Bromine Ionic Or Covalent The first question we ask is if the compound is ionic or covalent? Ionic bonds differ from covalent bonds. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located. Values are given for typical oxidation number and. Covalent radius half of the distance between two. Bromine Ionic Or Covalent.
From www.youtube.com
Is NH4Br (Ammonium bromide) Ionic or Covalent? YouTube Bromine Ionic Or Covalent Values are given for typical oxidation number and. That is, does it have ionic bonds, or covalent bonds?. Ionic compounds generally form from metals and nonmetals. However, in covalent bonds, the electrons are shared between. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in. Bromine Ionic Or Covalent.
From www.vrogue.co
Table Of Ionic Bonds Examples vrogue.co Bromine Ionic Or Covalent Compounds that do not contain ions, but instead consist of atoms bonded tightly. However, in covalent bonds, the electrons are shared between. Values are given for typical oxidation number and. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs of electrons that are located. That is, does it have. Bromine Ionic Or Covalent.
From www.slideserve.com
PPT PART I PowerPoint Presentation, free download ID2826467 Bromine Ionic Or Covalent However, in covalent bonds, the electrons are shared between. Covalent radius half of the distance between two atoms within a single covalent bond. Both types result in the stable electronic states associated with the noble gases. The first question we ask is if the compound is ionic or covalent? Ionic bonds differ from covalent bonds. Ionic compounds generally form from. Bromine Ionic Or Covalent.
From www.youtube.com
Is AlBr3 (Aluminum bromide) Ionic or Covalent/Molecular? YouTube Bromine Ionic Or Covalent Compounds that do not contain ions, but instead consist of atoms bonded tightly. Covalent radius half of the distance between two atoms within a single covalent bond. Values are given for typical oxidation number and. The first question we ask is if the compound is ionic or covalent? However, in covalent bonds, the electrons are shared between. Ionic compounds generally. Bromine Ionic Or Covalent.
From www.pastpapersinside.com
CHEMICAL BONDING Past Papers Inside Bromine Ionic Or Covalent Values are given for typical oxidation number and. That is, does it have ionic bonds, or covalent bonds?. Compounds that do not contain ions, but instead consist of atoms bonded tightly. Ionic compounds generally form from metals and nonmetals. Covalent radius half of the distance between two atoms within a single covalent bond. Covalent bonds are the attractive forces between. Bromine Ionic Or Covalent.
From fphoto.photoshelter.com
science chemistry covalent bonds Fundamental Photographs The Art of Bromine Ionic Or Covalent Values are given for typical oxidation number and. Ionic compounds generally form from metals and nonmetals. Compounds that do not contain ions, but instead consist of atoms bonded tightly. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. However, in covalent bonds, the. Bromine Ionic Or Covalent.
From www.numerade.com
Bromine is a halogen; one of the elements in the same column of the Bromine Ionic Or Covalent However, in covalent bonds, the electrons are shared between. Compounds that do not contain ions, but instead consist of atoms bonded tightly. Covalent radius half of the distance between two atoms within a single covalent bond. Both types result in the stable electronic states associated with the noble gases. Covalent bonds are the attractive forces between the positively charged nuclei. Bromine Ionic Or Covalent.
From www.chegg.com
Solved Si, Br ionic covalent Si, Br SiBr, silicon bromide Bromine Ionic Or Covalent Covalent radius half of the distance between two atoms within a single covalent bond. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. Covalent bonds are the attractive forces between the positively charged nuclei of the bonded atoms and one or more pairs. Bromine Ionic Or Covalent.
From edu.svet.gob.gt
How To Draw The Lewis Dot Structure For KBr Potassium Bromine Ionic Or Covalent That is, does it have ionic bonds, or covalent bonds?. Ionic compounds generally form from metals and nonmetals. Compounds that do not contain ions, but instead consist of atoms bonded tightly. However, in covalent bonds, the electrons are shared between. Values are given for typical oxidation number and. Ionic bonds differ from covalent bonds. The first question we ask is. Bromine Ionic Or Covalent.
From www.youtube.com
Reaction of Aluminium with Bromine YouTube Bromine Ionic Or Covalent Ionic bonds differ from covalent bonds. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. The first question we ask is if the compound is ionic or covalent? That is, does it have ionic bonds, or covalent bonds?. Both types result in the. Bromine Ionic Or Covalent.
From fphoto.photoshelter.com
science chemistry covalent bonds Fundamental Photographs The Art of Bromine Ionic Or Covalent Both types result in the stable electronic states associated with the noble gases. Values are given for typical oxidation number and. Covalent radius half of the distance between two atoms within a single covalent bond. Ionic bonds differ from covalent bonds. The first question we ask is if the compound is ionic or covalent? Compounds that do not contain ions,. Bromine Ionic Or Covalent.
From www.youtube.com
Draw the Lewis Structure of MgBr2 (magnesium bromide) YouTube Bromine Ionic Or Covalent Values are given for typical oxidation number and. However, in covalent bonds, the electrons are shared between. Ionic bonds differ from covalent bonds. Covalent radius half of the distance between two atoms within a single covalent bond. Ionic compounds generally form from metals and nonmetals. The first question we ask is if the compound is ionic or covalent? Both types. Bromine Ionic Or Covalent.
From www.youtube.com
How to Find the Valence Electrons for Bromine (Br) YouTube Bromine Ionic Or Covalent Both types result in the stable electronic states associated with the noble gases. Compounds that do not contain ions, but instead consist of atoms bonded tightly. The first question we ask is if the compound is ionic or covalent? That is, does it have ionic bonds, or covalent bonds?. Ionic bonds differ from covalent bonds. Covalent bonds are the attractive. Bromine Ionic Or Covalent.
From www.slideserve.com
PPT Ionic Bonding PowerPoint Presentation, free download ID2683450 Bromine Ionic Or Covalent The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the. Ionic bonds differ from covalent bonds. The first question we ask is if the compound is ionic or covalent? Values are given for typical oxidation number and. Ionic compounds generally form from metals and. Bromine Ionic Or Covalent.